1. Introduction
Water shortage has become a global issue nowadays due to the increase in industrialization and population to overcome such a problem water reuse and wastewater remediation are required
| [1] | Thirumoorthy K, Krishna SK. Removal of cationic and anionic dyes from aqueous phase by Ball clay - Manganese dioxide nanocomposites. J Environ Chem Eng. 2020; 8(1). https://doi.org/10.1016/j.jece.2019.103582 |
| [2] | Shabaan OA, Jahin HS, Mohamed GG. Removal of anionic and cationic dyes from wastewater by adsorption using multiwall carbon nanotubes. Arab J Chem. 2020; 13(3): 4797-4810. https://doi.org/10.1016/j.arabjc.2020.01.010 |
| [3] | Almoisheer N, Alseroury FA, Kumar R, Almeelbi T, Barakat MA. Synthesis of Graphene Oxide/Silica/Carbon Nanotubes Composite for Removal of Dyes from Wastewater. Earth Syst Environ. 2019; 3(3): 651-659. https://doi.org/10.1007/s41748-019-00109-w |
[1-3]
. The ecosystem is significantly impacted by the wastewater that various sectors produce. A serious environmental problem has arisen as a result of the world's antibiotic consumption rising quickly and the resulting discharge of pharmaceutical effluent. Antibiotics have serious mutagenic, genotoxic, and ecotoxicological impacts on ecosystems when they are introduced into wastewater
| [4] | Chakraborty P, Show S, Banerjee S, Halder G. Mechanistic insight into sorptive elimination of ibuprofen employing bi-directional activated biochar from sugarcane bagasse: Performance evaluation and cost estimation. J Environ Chem Eng. 2018; 6(4): 5287-5300. https://doi.org/10.1016/j.jece.2018.08.017 |
[4]
. Antibiotic-resistant genes, viruses, and bacteria in humans may arise as a result of antibiotics present in water bodies, raising grave concerns for public health
| [5] | de Carvalho JF, de Moraes JEF. Treatment of simulated industrial pharmaceutical wastewater containing amoxicillin antibiotic via advanced oxidation processes. Environ Technol. 2021; 42(26): 4145-4157. https://doi.org/10.1080/09593330.2020.1745296 |
[5]
. Because of their stubborn nature and resistance to degradation, the antibiotics are impervious to traditional wastewater treatment methods
. Consequently, the development of an efficient and effective treatment method for pharmaceutical wastewater containing antibiotics is imperative
| [7] | Cecconet D, Molognoni D, Callegari A, Capodaglio AG. Biological combination processes for efficient removal of pharmaceutically active compounds from wastewater: A review and future perspectives. J Environ Chem Eng. 2017; 5(4): 3590-3603. https://doi.org/10.1016/j.jece.2017.07.020 |
| [8] | Eryuruk K, Tezcan Un U, Bakır Ogutveren U. Electrochemical treatment of wastewaters from poultry slaughtering and processing by using iron electrodes. J Clean Prod. 2018; 172: 1089-1095. https://doi.org/10.1016/j.jclepro.2017.10.254 |
| [9] | Sher F, Malik A, Liu H. Industrial polymer effluent treatment by chemical coagulation and flocculation. J Environ Chem Eng. 2013; 1(4): 684-689. https://doi.org/10.1016/j.jece.2013.07.003 |
[7-9]
Wastewater from the pharmaceutical industry is poisonous and has a strong color and odor. The wastewater originating from the pharmaceutical industry presents challenges because of its high contents of organics (COD) and low values of BOD (biological oxygen demand). A complex network of organics is present in veterinary pharmaceutical wastewater
| [10] | Phoon BL, Ong CC, Mohamed Saheed MS, Show PL, Chang JS, LingTC, Jaun JC, Conventional and emerging technologies for removal of antibiotics from wastewater. J Hazard Mater. 2020; 400: 122961. https://doi.org/10.1016/j.jhazmat.2020.122961 |
[10]
. Such effluent may harm aquatic life and human health when it is released into the environment. Pharmaceutical effluent must be treated to comply with discharge regulations and save the environment
| [11] | Malik SN, Khan SM, Ghosh PC, Vaidya AN, Kanade G, Mudliar SN. Treatment of pharmaceutical industrial wastewater by nano-catalyzed ozonation in a semi-batch reactor for improved biodegradability. Sci Total Environ. 2019; 678: 114-122. https://doi.org/10.1016/j.scitotenv.2019.04.097 |
| [12] | Barzegar G, Wu J, Ghanbari F. Enhanced treatment of greywater using electrocoagulation/ozonation: Investigation of process parameters. Process Saf Environ Prot. 2019; 121: 125-132. https://doi.org/10.1016/j.psep.2018.10.013 |
[11, 12]
. One of the key stages in treating actual pharmaceutical wastewater is choosing a suitable treatment method. Information about the effectiveness of a specific strategy is crucial while doing that. Numerous scholars put up several methods for treating wastewater from pharmaceuticals. But these methods are less effective
.
Many treatment methods have been used to treat wastewater containing antibiotics, do not provide complete removal
| [14] | El-sayed MEA. Nanoadsorbents for water and wastewater remediation. Sci Total Environ. 2020; 739: 139903. https://doi.org/10.1016/j.scitotenv.2020.139903 |
| [15] | Rafique MS, Tahir MB, Rafique M, Khan MI. Chapter 13 - Recent advances in the development of photocatalysis and future perspectives. In: Tahir MB, Rafique M, Rafique MS, eds. Nanotechnology and Photocatalysis for Environmental Applications. Micro and Nano Technologies. Elsevier; 2020: 221-223. https://doi.org/10.1016/B978-0-12-821192-2.00013-9 |
[14, 15]
. While biological treatment methods like trickling filters
| [16] | Campos CJA, Avant J, Lowther J, Till D, Lees DN. Human norovirus in untreated sewage and effluents from primary, secondary and tertiary treatment processes. Water Res. 2016; 103: 224-232. https://doi.org/10.1016/j.watres.2016.07.045 |
[16]
and activated sludge
are environmentally friendly, they take a long time and only remove half of the antibiotics. Constructed wetlands function well, but they need a lot of space, and time to settle, and can't eliminate medications. High pharmaceutical removal can be achieved by physical techniques like adsorption
| [18] | Ihsanullah, Patel F, Khraisheh M, Atieh MA, Laoui T. Novel aluminum oxide-impregnated carbon nanotube membrane for the removal of cadmium from aqueous solution. Mater (Basel). 2017; 10(10). https://doi.org/10.3390/ma10101144 |
[18]
, however recycling and residue management are significant problems. Membrane filtration works well, although it is expensive and has a fouling issue
. Chemical methods
| [20] | Bernal-Martínez LA, Barrera-Díaz C, Solís-Morelos C, Natividad R. Synergy of electrochemical and ozonation processes in industrial wastewater treatment. Chem Eng J. 2010; 165(1): 71-77. https://doi.org/10.1016/j.cej.2010.08.062 |
[20]
for removing pharmaceuticals, such as ozonation
| [21] | Nemati Sani O, Navaei fezabady AA, Yazdani M, Taghavi M. Catalytic ozonation of ciprofloxacin using $γ$-Al2O3 nanoparticles in synthetic and real wastewaters. J Water Process Eng. 2019; 32(December 2018): 100894. https://doi.org/10.1016/j.jwpe.2019.100894 |
[21]
and sophisticated oxidation procedures, are very efficient, but they are expensive and energy-intensive. Hybrid treatment approaches can be used to get beyond the constraints of stand-alone treatments. Antibiotics are rapidly degraded by using a hybrid process that requires less reaction time and money to operate. The use of advanced oxidation processes (AOPs) for actual wastewater treatment has grown significantly for the past several years. For the treatment of several actual wastewaters, the catalytic ozonation procedure among AOPs showed superior performance
. Iron shavings were used in the catalytic ozonation process to treat effluent from bio-treated textiles. Another study effectively investigated the use of Nano-catalyzed ozonation for treating pharmaceutical wastewater. The color, toxicity, and COD were effectively removed by Nano catalyst (O
3/nZVI)
| [24] | Maher EK, O’Malley KN, Heffron J, Namara J, Wang Y, Mayer BK, HouJ. Analysis of operational parameters, reactor kinetics, and floc characterization for the removal of estrogens via electrocoagulation. Chemosphere. 2019; 220: 1141-1149. https://doi.org/10.1016/j.chemosphere.2018.12.161 |
[24]
. Even still, the analysis showed that the procedure was working more efficiently. However, the effectiveness of process depends upon several factors, including the catalyst's composition, the operational settings, and the effluents' characteristics. The effectiveness of the hybrid or combined electrocoagulation and catalytic ozonation procedure for treating basic yellow 28 dye in wastewater was examined in a few recent studies. However, application on real effluents were missing in the study. Various industrial effluents such as paint, textile, paper, pulp, distillery, home, etc. have been successfully treated by simple electrocoagulation methods due to electro-produced coagulants. Organic pollutants are eliminated by using in situ electro coagulants, which are generated during the anodic oxidation during the Electroflucculation process
| [25] | Mukimin A, Vistanty H, Zen N. Hybrid advanced oxidation process (HAOP) as highly efficient and powerful treatment for complete demineralization of antibiotics. Sep Purif Technol. 2020; 241: 116728. https://doi.org/10.1016/j.seppur.2020.116728 |
[25]
. Another study deals with the successful removal of carbamazepine, diclofenac, and amoxicillin by electrocoagulation processes from real municipal wastewater in an electrochemical reactor with an aluminum electrode. More than 50% of the antibiotics and 100% of COD were removed at the current density of 0.3 mA/cm, applied voltage 6 V and pH 7 in three hours.
| [26] | Ahmadzadeh S, Asadipour A, Pournamdari M, Behnam B, Rahimi HR, Dolatabadi M. Removal of ciprofloxacin from hospital wastewater using electrocoagulation technique by aluminum electrode: Optimization and modelling through response surface methodology. Process Saf Environ Prot. 2017; 109: 538-547. https://doi.org/10.1016/j.psep.2017.04.026 |
[26]
. The electrocoagulation process can be used to remove antibiotics from veterinary wastewater. In another study 88.57 % of the antibiotic removal at pH 7.78 and a current density of 12.5 mA/cm
2 after 20 minutes was achieved during electrocoagulation. Catalytic ozonation is an effective way to remove pollutants from wastewater.
According to the aforementioned research, stand-alone process may not be sufficient for treating real pharmaceutical wastewater due to the complex organic contaminants and the strong scavenging effect. As a result, a high removal efficiency of 97.8 % was recorded after 80 minutes. with a catalyst dosage of 0.1 g and an ozone dose of 2 mg/L. The effectiveness of Fe shavings in the process for eliminating sulfamethoxazole from real municipal wastewater was analyzed. At pH 7, an ozone dose of 178.8 mg/L, an iron quantity of 288.46 g/L, and rapid 99 % removal of antibiotics was achieved within 5 min. COD elimination was recorded as 80% at 60 minutes. In another study published a method of treating antibiotics such as amoxicillin and acetaminophen by combined ozonation and adsorption process with cross-linked chitosan/bentonite. The combination process at pH 7 and an ozone dose of 15 mg/L provided complete removal. Pharmaceutical wastewater has been treated successfully by combining catalytic ozonation and Electroflucculation process would be the most effective for reducing the levels of pharmaceutical antibiotics in wastewater, as previously reported
| [27] | Jaafarzadeh N, Barzegar G, Ghanbari F. Photo assisted electro-peroxone to degrade 2, 4-D herbicide: The effects of supporting electrolytes and determining mechanism. Process Saf Environ Prot. 2017; 111: 520-528. https://doi.org/10.1016/j.psep.2017.08.012 |
[27]
. According to published research, AOPs are useful alternative methods for degrading organic pollutants. Their methodology involves generating free OH radicals as oxidizing agents to facilitate the mineralization of complex substances found in wastewater. Hydroxyl radicals (•OH) have a 2.80 V oxidation potential, making them powerful oxidizing agents that undergo faster oxidation processes than traditional oxidants like potassium permanganate and hydrogen peroxide.
UV and ozone have long been employed in wastewater treatment and water systems to prevent the growth of pathogenic organisms. Comparably, ozone is a potent oxidizing agent that combines with aromatic and unsaturated moieties in a targeted manner to quickly decolorize. Because hydroxyl radicals have a greater capacity for oxidation than ozone (O
3) alone, the combined ozone/UV process can efficiently destroy organic contaminants. When ozone and UV light interact in an aqueous solution, the ozone interacts with the water to create hydroxyl radicals, or •OH radicals
| [27] | Jaafarzadeh N, Barzegar G, Ghanbari F. Photo assisted electro-peroxone to degrade 2, 4-D herbicide: The effects of supporting electrolytes and determining mechanism. Process Saf Environ Prot. 2017; 111: 520-528. https://doi.org/10.1016/j.psep.2017.08.012 |
[27]
. The following reactions show that they are less selective and stronger than any other kind of oxidant.
The prime parameters that may be responsible for the progress of ozone/UV systems are UV irradiation level, pH, irradiation level, and ozone dose. Photoassited Ozonation is commonly used for the oxidation and effective degradation of toxic compounds that may present in wastewater. In the photo-assisted ozonation process, the oxidizing potential of ozone is accelerated due to the formation of free •OH radicals. A combined AOPs for the effective removal of sulphamethoxazole showed a high removal during the UV-ozonation process
| [28] | Process OE, Javaid R. Treatment of Textile Wastewater by a Novel Clay/TiO 2 /ZnO- Based Catalyst, Applying a Synergic Catalytic Ozonation–Electroflocculation Process. 2023. |
[28]
. The titania and zinc oxides are cheap and non-toxic photocatalysts that have been widely used for wastewater treatment however large band energy gap and separating issues limited their usage on a large commercial scale that’s why immobilizing these photocatalysts on clay have removed this issue along with enhanced adsorption capacities and removal efficiencies. A study has been made on treating real textile effluents by clay/titania/ZnO using combined ozonation and Electroflucculation process however the effectiveness of this photocatalyst is never tested under UV-C lamp and for antibiotics removal from real pharmaceutical wastewater
| [29] | Mahmood Q, Shaheen S, Bilal M, Ali A., Zeb BS, Tariq M. Chemical pollutants from an industrial estate in Pakistan: a threat to environmental sustainability. Appl Water Sci. 2019; 9(3): 1-9. https://doi.org/10.1007/s13201-019-0920-1 |
[29]
. This study investigated their photolytic activity for antibiotic degradation under real conditions.
In this study combine UV with Electroflucculation and catalytic ozonation approach has been used to remove amoxcyline and carbamazepine from veterinary pharmaceutical wastewater using a novel photocatalyst clay/TiO2/ZnO. Since titanium (TiO2) and zinc oxide (ZnO) are photocatalysts, their photocatalytic activity is enhanced under UV. According to Arthur’s Knowledge, this approach with a UV lamp has not been used previously studied for the removal of antibiotics from real pharmaceutical wastewater using Clay/TiO2/ZnO. Additionally, in the current study, the mechanism of turbidity and COD reduction from actual pharmaceutical wastewater was investigated, along with the effects of several operational parameters like pH, DC voltage, catalyst dose, and ozone dose.
2. Methods and Method
2.1. Materials
The Novamed Pharmaceuticals pharmaceutical firm, situated on Kasur Road in Lahore, Pakistan, provided the actual veterinary pharmaceutical wastewater. The wastewater was collected, brought to the lab, and kept there right away at 4°C. Before the treatment method, the sample was described.
Table 1, provides the initial characterization of wastewater containing veterinary pharmaceuticals. The following substances and reagents were employed to calculate the COD: sulfuric acid, starch, pH, potassium iodide, and NaOH. The reagents and chemicals were all of analytical grade.
Table 1. Characterization of Pharmaceutical Wastewater.
Parameters | Before treatment | After treatment | NEQS 30] |
pH | 10 | 7 | 6-9 |
Turbidity | 289 NTU | 53 NTU | - |
COD | 1600 mg/L | 100mg/L | 150 mg/L |
BOD | 580 mg/L | 50 mg/L | 80 mg/L |
2.2. Catalyst Preparation
Commercial ZnO (P25) and TiO
2 (P25) powder were acquired from Merck Germany. Using the incipient impregnation procedure, 5 M sulfuric acid was added to bentonite (natural montmorillonite, >2 µm) to generate the ternary composite. For different contact hours, this required combining 200 g of bentonite clay with a 1 L aqueous media of 5 M sulfuric acid (271 mL) at 80 °C. After that, the mixture was washed with demineralized water to bring its pH down to zero. Next, the dye solution was dried in the oven at 105°C to correct its pH. Subsequently, distilled water was used to mix the 50% w/w acid-treated bentonite clay, 50% w/w TiO
2, and 5% w/w ZnO. These mixtures were then placed in a sonicator (100 W, 40 KH, universal ultrasonic DSA-100-SK1-2.8 L stainless-steel Cleaner) to form a uniform mixture. After that, this mixture was dried for twenty-four hours in a conventional oven and then calcined for three hours at 660 °C (heating rate: 10 °C/min) to remove any remaining impurities
.
2.3. Experimental Procedure
The experimental assembly used in this work is shown in
Figure 1. The two aluminum flat electrodes were separated by a 20 cm distance and housed in a rectangular borosilicate glass tank. The electrodes' effective area measured 41.23 cm³. The ozone generated by the ozone generator was able to bubble through the Sparger (diffuser) located at the bottom of the reactor. The electricity was produced by the DC power supply. The UV-C lamp (36-watt bulb, 508 mm length) used in this experiment emits monochromatic radiations at 254 nm. There was UV radiation of 1.2 mW/cm
2. Every run started with the placement of a one-liter wastewater sample in the reactor, followed by the adjustment of the pH, the determination of the necessary ozone dose and voltage, and the collection of residue samples for analysis.
Figure 1. Schematic Representation of Hybrid O3/UV/EF Reactor setup.
2.4. Ozone Dose Analysis
Using the standards idometric method, the inlet ozone dosage supplied to the reactor was calculated
| [32] | Ikhlaq A, Aslam T, Zafar AM, Javed F, Munir HMS. Combined ozonation and adsorption system for the removal of heavy metals from municipal wastewater: Effect of COD removal. Desalin Water Treat. 2019; 159: 304-309. https://doi.org/10.5004/dwt.2019.24164 |
[32]
. For a predetermined amount of time, gas bottles containing a 2 % KI solution were filled with the O
3 gas being generated by the ozone generator (30 min). The sample was quenched by using a 2 N H
2SO
4 solution. Using a starch indicator, it was titrated against 0.005 Normal sodium thiosulphate solution (Na
2S
2O
3) in the following step. The ozone dose was computed using the following formula.
Ozonedose(mg/min)=
Ozonedose(mg/min)=
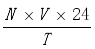 (1)
(1) Here, V is the Volume of titrant used and N is the Normality of Na2S2O3.
2.5. Chemical Oxygen Demand (COD)
The initial (COD
i) and final COD (COD
f) concentrations of the raw and treated samples were determined using the traditional HACH reactor digestion method (8000)
| [33] | Assila O, Tanji K, Zouheir M, et al. Adsorption studies on the removal of textile effluent over two natural eco-friendly adsorbents. J Chem. 2020; 2020. https://doi.org/10.1155/2020/6457825 |
| [34] | Masood Z, Ikhlaq A, Farooq U, Qi F, Javed F, Aziz HA. Removal of anti-biotics from veterinary pharmaceutical wastewater using combined Electroflocculation and Fe-Zn loaded zeolite 5A based catalytic ozonation process. J Water Process Eng. 2022; 49(May): 103039. https://doi.org/10.1016/j.jwpe.2022.103039 |
[33, 34].
CODremovalefficiency[%]=(
CODremovalefficiency[%]=(
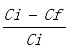
)×100
(2) 2.6. Pharmaceuticals Analysis
The pharmaceuticals were measured with the HPLC equipment (L-2130, japan, Hitachi Elite Lachrom) connected to a (4.6 × 250 mm) C18 column (Poroshell 120)
| [35] | Ikhlaq A, Zafar M, Yasir A, Akram A, Shabbir S, Qi F. Catalytic ozonation for the removal of reactive black 5 (RB-5) dye using zeolites modified with CuMn2O4/gC3N4 in a synergic electro flocculation-catalytic ozonation process. Water Sci Technol. 2021; 84(8): 1943-1953. https://doi.org/10.2166/wst.2021.404 |
[35]
. The wavelength was set to 250 nm, and the mobile phase consisted of methanol and deionized water in 50: 50 ratios, respectively. A 10 μL injection volume was used, with a 1 mL/min flow rate, and a 254 nm wavelength. Using an internal standard procedure, the unidentified drugs were found in the veterinary effluent. For amoxicillin, the limits of detection and quantification were measured and determined to be 20 μg/L and 80 μg/L, and for carbamazepine, 30 μg/L and 100 μg/L, respectively. Using the following methodology, the % removal efficiencies of pharmaceuticals were calculated from the peak areas in the sample and the peak areas post-treatment.
PharmaceuticalRemoval(%)=
PharmaceuticalRemoval(%)=
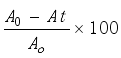 (3)
(3) 3. Results and Discussion
3.1. Voltage Optimization
The progress of the combine and single process strongly depends on the applied DC voltage. The effect of voltage on the turbidity and COD reduction form actual pharmaceutical wastewater was investigated at three different levels of 5-15 V.
Figure 2 (a, b). By increasing the electrolysis time and the applied voltage, increased both turbidity and COD removal. The COD removals of 44%, 86.9%, and 89.3% were achieved after one hour at 5 V, 10 V, and 15 V respectively. Moreover, an abrupt decline in the turbidity by increasing the applied voltage was observed in the presence of UV. The turbidity values decline to 59 NTU, 75 NTU, and 110 NTU after one hour at applied DC voltages from 15 V-5 V respectively. This may be because enhancing the applied voltage the ions generated by the electrode dissolution increase. This result in producing various species of metal hydroxides causing charge neutralization of organics present in the raw water causes ultimate removal via sweep coagulation/flocculation
| [36] | Munir HMS, Feroze N, Ikhlaq A, Kazmi M, Javed F, Mukhtar H. Removal of colour and COD from paper and pulp industry wastewater by ozone and combined ozone/UV process. Desalin Water Treat. 2019; 137: 154-161. https://doi.org/10.5004/dwt.2019.23186 |
| [37] | Duan X, Wu P, Pi K, Zhang H, Liu D, Gerson AR. Application of Modified Electrocoagulation for Efficient Color Removal from Synthetic Methylene Blue Wastewater. 2018; 13: 5575-5576. https://doi.org/10.20964/2018.06.15 |
[36, 37]
. From
Figure 2 (a, b) we can easily depict that the COD removal efficiency after one hour was not as significantly high at 15 V than at 10 V. because under UV-assisted electrochemical process, the consumption of energy significantly increases with the increase in voltage. The 10 V was selected as an optimum voltage, due to economic aspects, and more than 85 % of turbidity and COD removal was obtained. However, by increasing voltage, under UV, there is an increase in the production of free
•OH radicals. The O
2 reduction leading to the generation of H
2O
2 which accelerates the O
3 conversion to free • OH radicals, thereby increasing the removal of the organic. Furthermore, for the ozone/UV process, (OH) radicals are surrounded by water clusters approach the cage effect. The H
2O
2 being generated reacts with O
3 producing more free •OH radicals. The photo-assisted process can significantly degrade the organics because the OH radicals have a higher oxidizing potential than ozone (O
3) alone. In the ozone/UV process the reactions in aqueous media, the O
3 energizes and reacts with water to form free (•OH radicals) that are less selective than other types of oxidants
| [38] | Javed F, Ahmad SW, Ikhlaq A, Rehman A, Saleem F. Elimination of basic blue 9 by electrocoagulation coupled with pelletized natural dead leaves (Sapindus mukorossi) biosorption. Int J Phytoremediation. 2020; 0(0): 1-12. https://doi.org/10.1080/15226514.2020.1825328 |
[38]
.
3.2. Effect of pH
The organic compound’s reaction pathway of the combined process is highly affected by the pH. EF is normally a self pH neutralization process which initially increase the pH under the basic range. This happens due to water electrolysis, generation of OH
− ions, and H
2 evolution. At pH = 7 the stability in the EF process can be seen due to Aluminum (Al) conversion and formation of insoluble aluminum hydroxides. On the other hand, ozonation is a pH-driven process, favoring the radical-based mechanism by producing free •OH radicals under alkaline pH ranges and direct molecular (O
3) in acidic pH ranges. For the successful removal of organics for catalytic ozonation, the surface charge of nanocatalyst is governed by the reaction kinetics. In the presence of UV light, the turbidity and COD removal was investigated for actual pharmaceutical wastewater at pH values of 5, 7, and 10. The findings displayed in
Figure 3 demonstrate noticeably improved turbidity and COD reductions with pH. After 60 minutes at pH 10, 7, and 5, respectively, the COD reduction increased to 90.1%, 86.9%, and 69.2%, and turbidity values of 59.0 NTU, 74.0 NTU, and 90.2 NTU were attained. Because there were more free radicals under acidic pH, the COD elimination efficiency rose to more than 85% when the pH was raised to 5-7.
The maximum reductions achieved at pH 10 (53 NTU and 86.9% COD removal) in
Figure 2 show the presence of highly cationic organic pollutants in the wastewater. Under alkaline pH 10, the combined effect of UV/catalytic ozonation and Electroflucculation/UV greatly enhanced the organics abatement. The O
3/UV process showed better performance under alkaline conditions for turbidity and COD as shown in
Figure 2 (c, d). due to the result of fast organic reactions with free hydroxyl radicals. At alkaline pH Furthermore, in the case of the ozone/UV process, hydroxyl radicals that are surrounded by an H
2O cluster may approach each other, and ozone which is a self-decomposition to radicals starts as a result of initiation reaction. by the cage effect and H
2O
2 is produced that reacts with O
3 producing more free •OH radicals.
The most prevalent species in the Electroflucculation process at the higher pH level is insoluble aluminum hydroxide
| [20] | Bernal-Martínez LA, Barrera-Díaz C, Solís-Morelos C, Natividad R. Synergy of electrochemical and ozonation processes in industrial wastewater treatment. Chem Eng J. 2010; 165(1): 71-77. https://doi.org/10.1016/j.cej.2010.08.062 |
[20]
. the self-decomposition was accelerated by O
3 to produce radicals. The abundance of negatively charged surfaces on the catalyst and OH radicals, which quickly break down organics through radical mechanisms are all variables that lead to the accelerated breakdown of organics at pH 10 in comparison to other pH levels that have been investigated. Furthermore, at higher pH levels, the system's ozonation bubbling and the synergistic dual effect of the fine hydrogen bubbles generated in the EF process greatly increase turbulence, mixing, and gas-solid-liquid interface contact with increased ionic mobility, resulting in a rapid organics abatement. In the current investigation, the highest COD reduction of 90.1 percent was attained at pH 10 (
Figure 2 (c)), whereas 86.9 percent was attained at pH 10. The experiments were conducted under real conditions, without adjusting the pH, because the actual pharmaceutical wastewater was obtained at pH 10. This was done because it may not be cost-effective to change the pH of wastewater before treating it on a larger scale in practical applications. Thus, pH 10 was chosen as the optimum pH for the current experiment. displayed in
Figure 2 (c, d).
Figure 2. (a, b) Effect of Voltage on % removal of COD and Turbidity at pH10, Ozone dose 0.6 mg/min, Voltage 10V, catalyst dose 1 g/L, UV 1.2 Mw cm-2 (c, d). Effect of pH on % removal of COD and Turbidity, at voltage 10 V, Ozone dose 0.6 mg/min, catalyst dose 1 g/L, and UV 1.2 Mw cm-2.
3.3. Effect of Catalyst Dosage
The catalyst plays a crucial role in the hybrid Electroflucculation/UV/catalytic ozonation process because of its potent adsorption capability and kinetics for the organics abatement organics from actual pharmaceutical wastewater. The relevant photocatalytic activity of the combined process was examined by varying the catalytic dose between 0.5 and 1.5 g/L and noting the discernible drops in turbidity and COD of the effluent. The catalytic activity is validated by the data in
Figure 4, which shows a favorable connection between catalyst dose and decreases in COD and turbidity. Turbidity values decreased to 220 NTU and COD removal increased from 34.2 percent to 90.1 percent with an increase in catalyst dose from 0.5 g/L to 1.5 g/L. Given that over 85% of the removal was attained at a dose of 1 g/L, we regard 1 g/L as the ideal dosage for economic reasons. Better removal offered by the catalyst dosage is due to the accessible surface area, porosity, adsorption capacity, and kinetics, as illustrated in
Figure 3.
Figure 3. Effect of Catalyst at pH10, Ozone dose 6 mg/min, UV 1.2 Mw/cm-2, Voltage 10 V.
3.4. Effect of Ozone Concentration
The combined UV/Electroflucculation/ozonation process produces oxidants. when the concentration of ozone and O
2 mixture in the solution is increased, causes a significant increase in treatment efficiency. As shown in
Figure 4, the ozone dose highly improves the turbidity and COD reduction based on actual pharmaceutical wastewater. An ozone dose of 0.2 mg/min, 0.6 mg/min, and 0.8 mg/min was utilized to achieve COD removal rates of 80 percent, 83.4 percent, and 85.4%, respectively. Because an increase in (O
3) gas directly accelerates diffusion via the gas-liquid interface, the concentration of dissolved ozone rises to more readily attack organic molecules. Radical-based reactions happen on the surface of the catalyst at higher rates when ozone levels are higher. Since raising the ozone dose from 0.6 mg/min to 0.8 mg/min did not significantly alter the situation, 0.6 mg/min was determined to be the ideal amount. Nonetheless, the measurements of turbidity were 98 NTU, 85 NTU, and 80 NTU, in that order.
Figure 4. Effect of Ozone dose at pH10, voltage 10 V, UV 1.2 mW/cm-2.
3.5. Catalyst Reuse Performance
The photocatalyst was cleaned, heated, and dried according to the recommended method to assess the catalyst's reusability. The fourth cyclic run (
Figure 5 (a, b)) showed a decline in turbidity removal up to 132 NTU and COD removal efficiency of up to 56%, which may have been caused by the organics covering the porous surface of the catalyst. The catalyst surface continuously auto-regenerates in the UV/Electroflucculation/catalytic ozonation combined process as a result of the hybrid mechanism of O
3 oxidation, photo-generated electrochemical oxidation, or the oxidation of the in-situ generated radical. This preserves the catalyst's sorption capacity and increases catalyst life and replacement needs. Because organic contaminants cover the porous surface of the nanocomposite, these calculations verified that the material is stable enough for long-term use under real-world settings. as well as to improve catalyst life. The photocatalyst's surface contained a small amount of titania, which reacted with the solution's accessible OH- ions over time to generate titanium hydroxides (H
4O
4Ti
3). These hydroxides functioned as coagulants and flocculants, increasing the removal efficiency. Because the H
4O
4Ti
3 produced by the Aluminum electrode in the hybrid batch reactor is so little, the production of aluminum hydroxides was thought to be a major coagulant. As a result, following each experimental run, a thin layer of sludge developed at the surface as a result of aluminum hydroxides (AL(OH)
3) brought on by oxygen produced at the anode floating there.
Figure 5. (a, b) Catalyst Reusability for COD and Turbidity removal at pH10, Ozone dose 0.6 mg/min, Voltage 10 v, UV 1.2 mW/cm-2, Catalyst dose 1 g/L.
3.6. Comparison of all Processes with and Without UV
The results shown in
Figure 6 (a, b) % COD and turbidity reduction were maximum for the combined process than other studied processes. The removal efficiency order was Combined UV > catalytic ozonation-UV, ozonation-UV, and Electroflucculation-UV process compared to the sole process. This may be because the photo-assisted ozonation/Electroflucculation process studied at pH (pH = 10) promotes the fast generation of free OH radicals that have a higher removal rate than oxidant (O
2) generated in the Electroflucculation/UV or simple electrolysis process. For turbidity reduction, however, the photo process was considered to be more efficient than others. Moreover, interestingly processes involving photo-assisted Electroflucculation (EF/UV) were shown higher turbidity reduction than others. This could be a result of the aluminum hydroxide flocs generated during Electroflucculation (EF), which absorbed the suspended particles in the wastewater. Consequently, the outcomes validate that treating actual wastewater with a combined approach would be a superior choice. While the O
3/UV procedure improved color removal from 80 percent and 81 NTU at pH 10, COD removal efficiency and turbidity were reported at 78.9 percent and 100 NTU at pH 10. The greater removal efficiency achieved in the UV - ozonation process. UV light produces fast (•OH) radicals during interaction, while O
3 also produces more hydroxyl radicals under basic conditions that result in a faster reaction between radical species and organic matters. In the presence of photo-assisted catalyst the COD and Turbidity removal further boost to 83.4% and 68 NTU. Due to the surface area and porosity of the nanocatalyst since the catalyst is a photocatalyst the catalytic activity enhanced in the presence of UV
Figure 6. (a, b) as compared to UV as 80% and 89 NTU in
Figure 6 (a, b). The combined photo/EF/O
3 process provides the maximum removal of COD and Turbidity at 86.9% and 81% compared to the alone synergic process without UV at 82% and 78% due to the fast generation of free OH radicals.
Figure 6. (a, b). Comparison of all process for COD and Turbidity removal at pH10, Ozone dose 0.6 mg/min, Voltage 10 V, UV 1.2 mW/cm-2, Catalyst dose 1 g/L.
3.7. Pharmaceuticals Removal
The findings shown in
Figure 7 the synergic procedure was highly effective in eliminating both of the identified drugs from wastewater. When amoxicillin was compared to other substances, it was shown that its % removal was the highest. For instance, the clearance efficiency of amoxicillin was 100% while that of carbamazepine was approximately 95%. According to
Table 1's results, wastewater can be discharged into waste streams with confidence because the effluents meet all national environmental quality parameters (NEQs) within a reasonable range. It is crucial to note that for wastewater to be recycled, it must be useful and meet certain requirements. For instance, to be used for irrigation, some extra requirements may apply, such as the water's salinity. The goal of the present study was to determine whether the synergistic process in a batch hybrid reactor is effective. It was discovered that, in terms of the parameters examined, treated real pharmaceutical wastewater lies within the NEQs.
Figure 7. Pharmaceutical removal at pH10, Ozone dose 0.6 mg/min, Voltage 10 V, UV 1.2 mW/cm-2, Catalyst dose 1 g/L.
3.8. Proposed Mechanism
Figure 8 illustrates the suggested mechanism of the combined process for the removal of COD and organic contaminants. The findings demonstrated that the synergistic process accelerated the rate of decomposition of the organic contaminants, which led to a drop in COD and color. At pH 10, the greatest amount of organic contaminants was eliminated due to side-by-side oxidation mechanisms sweep coagulation/flocculation. The UV/catalytic ozonation, and Electroflucculation occurred simultaneously in a modified hybrid reactor. Free hydroxyl radicals are produced at the anode during the electrolysis-UV process of producing hydrogen peroxide. While a reduction at the cathode results in OH radicals, oxidation at the anode promotes the production of O
2 and metal ions. Through destabilization and neutralization, the contaminants absorb the aluminum flocculation. Both direct and radical oxidation processes for ozonation were present at pH 10. By a variety of potential methods, bentonite supplied the platform for the quick production of free OH radicals in a solution. Moreover, the nanocatalyst's due to adsorption capacity supplied the surface for the quick production of hydroxyl (•OH) radicals, which eliminated the organic pollutants by converting O
3 to O
3− and O
2− and generating more hydrogen peroxide (H
2O
2), which in turn caused the organic pollutants to leach. Ozonation-UV and EF-UV exhibited better performance than simple ozonation and electrolysis. Ozonation in combination with UV and Catalyst was more effective since high amounts of OH radicals were produced through activation of O
3.
Figure 8. Proposed mechanism for combined process.
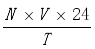 (1)
(1) 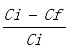 )×100(2)
)×100(2) 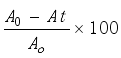 (3)
(3)